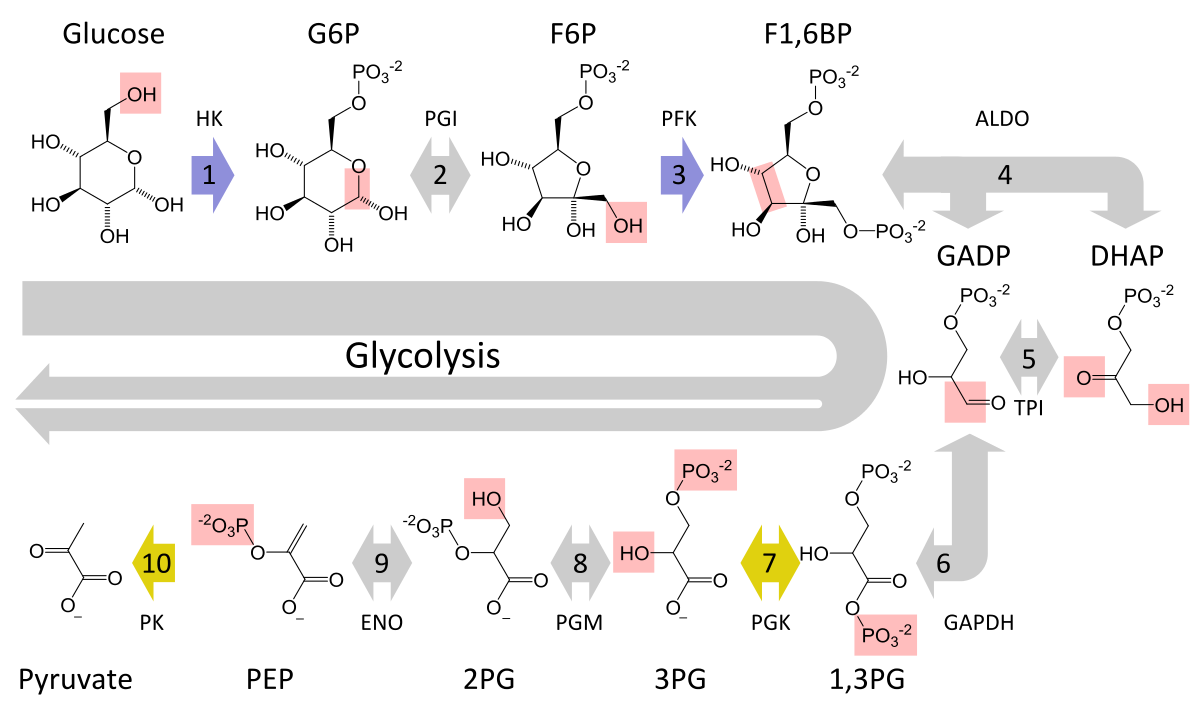
The Ten Steps of Glycolysis
Glycolysis (glycol: “sugar”, lysis: “to split”) is the process by which a six carbon glucose molecule is broken down, or catabolized, into a pair of three carbon pyruvate molecules. Doing so releases energy in the form of ATP. Pyruvate, meanwhile, serves as a precursor in many other metabolic pathways including fermentation and the citric acid cycle.
Glycolysis is thought to be one of the oldest metabolic pathways since it does not require oxygen (anaerobic), consistent with the conditions during the first billion years on earth. But also because its occurs in most living organisms (there are a few microorganisms which lack the glyositic enzymes). In every organism for which it does occur, glycolysis takes place in the cytoplasm, outside the mitochondria (if there is one). It does so anaerobically.
Glycolysis will continue into fermentation in anaerobic conditions, such as an active muscle, producing lactate. Another form of fermentation that occurs in yeast and bacteria produces ethanol.
In the aerobic conditions of the mitochondria, pyruvate is completely degraded into H2O and CO2 during the citric acid cycle and oxidative phosphorylation. This total oxidation of glucose is highly exergonic, releasing 2480 kJ of energy. Though it is a spontaneous reaction, without enzymes it would be impossibly slow–so, there’s no risk of a bowl of sugar spontaneously exploding in your kitchen! During glycolysis, enzymes speed the process along in a series of ten steps. These enzymes also controls the release of energy.
The ten steps of glycolysis are divided into two phases, the investment (or preparatory) phase and the payoff phase, so named based on whether they require or produce ATP. The required enzyme is shown in parenthesis.
The investment phase
1. Glucose —(hexokinase)—> Glucose-6-Phosphate (G6P)
cost: ATP, gain: ADP
One of the interesting side effects of phosphorylating glucose is that cell membranes lack a transporter to export G6P due to its negative charge. Thus, once phosphorylated, it remains trapped inside the cell. The phosphorylation also serves to destabilize Glucose, which helps further its metabolism.
Step one is an irreversible step, which becomes relevant when discussing the reverse of glycolysis, glyconeogenesis. Also of interest: in the liver, as it’ll be shown, an isomer of hexokinase called glucokinase serves as the enzyme for this step.
2. G6P —(phosphoglucose isomerase)—> Fructose-6-Phosphate (F6P)
This is an isomerizing step. The atoms get shuffled around in the G6P molecule. Specifically, the G6P is linearized, its carbonyl group moves from C1 to C2, then it gets cyclized into F6P.
3. F6P —(phosphofructokinase, a.k.a., PFK-1)–> Fructose-1,6-diphosphate (F1,6P)
cost: ATP, gain: ADP
Now that the C1 has been relocated, it’s primed to be phosphorylated by an ATP molecule. Doing so produces a Fructose with two phosphate groups. This destabilizes the molecule by bringing two negatively charged phosphate groups in close proximity, thus priming the cleavage that follows…
Step 3 is an incredibly important step for other pathways/processes, as will be shown in later sections. This is also an irreversible step.
4. Split step!
F1,6P —(aldolase)–> dihydroxyacetone phosphate (DHAP)
F1,6P —(aldolase)–> Glyceraldehyde-3-phosphate (G3P)
The six carbon Glucose is cleaved into two three carbon molecules here, DHAP and G3P. The aldolase linearizes the F1,6P before the cleaving. DHAP will be converted to G3P in step 5 putting both on the same path again.
5. DHAP —(triosephosphateisomerase)—> Glyceraldehyde-3-phosphate (G3P)
So now the two three carbon molecules are back on the same path…
Sidebar: Triosephosphateisomerase, a.k.a., TIM, is a remarkable enzyme. First, it speeds the reaction 10^10 fold. Think one second versus 300 years. Furthermore, TIM consumes all of its substrate and is only rate-limited by how fast molecules can diffuse to it. As such, it’s considered a “kinetically perfect” enzyme.
This is an interesting example of Le Chatelier’s Principle too. Technically, TIM converts DHAP -> G3P. But because we’re using all of the G3P, the concentration drives it the other way
The payoff phase
For steps 6-10, double all costs and gains since it happens twice per glucose
6. G3P —(triosephosphate dehydrogenase)—> 1,3-biphosphoglycerate (1,3-BP)
cost: NAD+, P, gain: NADH, H+
Here, the phosphorylation of G3P into 1,3-BP comes from a stray phosphate ion, coupled with an NAD+, electron acceptor. This helps prime it for the hydrolysis of ATP that follows.
This step has varied outcomes in red blood cells, which will be explained in detail later.
7. 1,3-BP —(phosphoglycerokinase)—> 3-phosphoglycerate
cost: ADP, gain: ATP
Releases a phosphate. This form of ATP synthesis is called substrate-level phosphorylation.
8. 3-phosphoglycerate —(phosphoglyceromutase)—> 2-phosphoglycerate
This is mutation step. The atoms just get shuffled around in the 3P molecule.
9. 2-phosphoglycerate —(enolase)—> Phosphoenolpyruvate (PEP).
gain: H20
The reason phosphoglycerate doesn’t donate its phosphate directly to ADP to form ATP is because the free energy of hydrolysis of PEP is much higher (~3.5x) than the free energy of phosphoglycerate. Thus, it’s more energetically favorable to have it happen in the last step instead of here.
10. PEP —(Pyruvate kinase)—> Pyruvate.
cost: ADP, gain: ATP
Releases a phosphate. This is also an irreversible step. Thus ends glycolysis!
Score card
Inputs: 4 ADP, 2 Pi, 2 NAD+, 2 ATP
Outputs: 2 Pyruvate, 4 ATP (so net of 2), 2 NADH, 2 H+, 2 H20
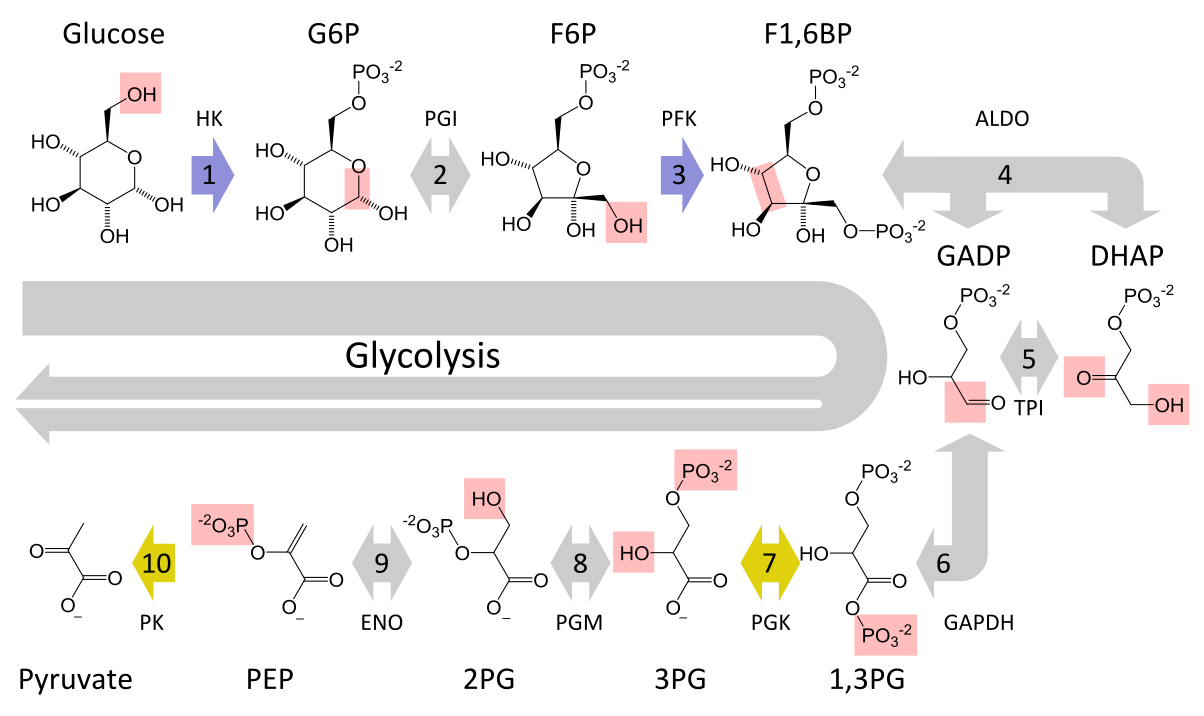
Note: steps 1, 3, and 10 are marked irreversible. This will be very important in the discussion of glyconeogenesis.